Nuclear fission occurs when a large atomic nucleus is split by means of neutron bobardment into smaller nuclei.
Where the energy comes from in Nuclear Fission?
Why use Uranium for Nuclear Fission?
Difficulties with nuclear fission as an energy source
Nuclear Fission and the environment
Plutonium Fast Breeder Reactors
Nuclear Fission
Nuclear fission occurs when an atomic nucleus is split into smaller nuclei. Unlike Radioactive decay, which is spontaneous and not considered a nuclear reaction, nuclear fission must be initiated by neutrons.
Where the energy comes from in Nuclear Fission?
With the exception of hydrogen –which only contains a proton in its nucleus – all the elements are less massive than the mass of their constituent parts. For example helium contains four nucleon, 2 protons and 2 neutrons, in its nucleus. If you had added the mass of the individual nucleons together they would have more mass than a helium nucleus. So where has the mass gone? The missing mass has become the energy required to hold the nucleus together (the strong nuclear force) and this energy is known as the binding energy.
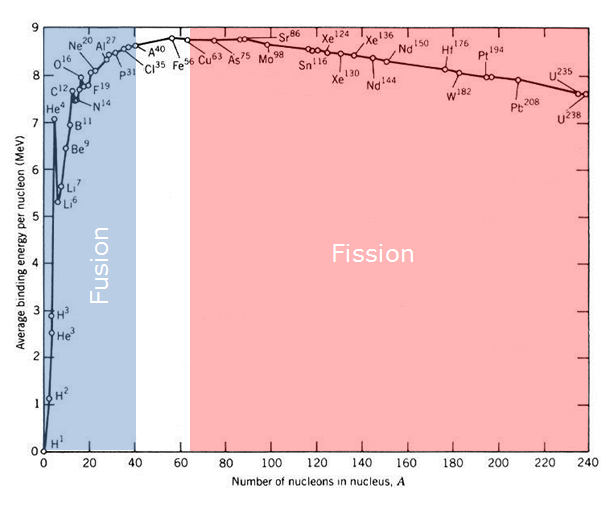
The table above shows the amount of binding energy in each nucleon of each element. The higher binding energy per nucleon the more stable the nucleus of the element is and all elements want to be as stable as possible. As you can see from the table Iron is the most stable (has the most binding energy per nucleon) element and as such neither nuclear fusion nor fission is possible. As you can see from the table, all elements with less mass then iron will release energy through nuclear fusion as these element gain nuclear binding energy per nucleon as their mass increases. Elements with more mass than iron have less binding energy per nucleon and so they release energy from splitting into less massive elements through nuclear fission.
Induced Nuclear Fission
It is possible to produce energy through the process of nuclear fission. This energy production is utilised in nuclear bombs and nuclear power stations. Nuclear power stations generally strike a nucleus of Uranium or plutonium using neutrons with the aim of causing the nucleus to split. Sometimes in neutron bombardment the neutron will actually combine making the uranium unstable which causes the nucleus to split but normally it is the force of the neutron that causes the fission to occur.
Nuclear Fission Reactors
In nuclear reactors stable uranium or plutonium is bombarded with neutrons to induce nuclear fission. Striking a stable uranium atom with a free neutron can change the uranium into an unstable isotope of uranium. This unstable uranium will then split through radioactive decay producing two smaller atoms such as Krypton and Barium, energy (about 210 MeV) and even more neutrons. These neutrons are propelled out and will collide with another uranium nucleus causing a chain reaction. Each time energy is released, and it is quite possible for the chain reaction to get out of control if left to its own devices. To control the output and prevent too much energy release a neutron absorbing element is introduced. In reactors control rods are introduced which are made of boron or cadmium and absorb many of the free neutrons. The perfect ratio is that one of the free neutrons released hits another nucleus and this is said to be at critical state. If too few free neutrons are hitting the uranium 235 neutron, and instead being absorbed by either the uranium-238 or reactor casing etc, then the reaction is said to be sub critical. If more than one free neutron per fission strike a uranium-235 atom, and the chain reaction grows, the reaction is said to be supercritical and the rods can be lowered until criticality is reached once more.
The animation shows the reaction when a free neutron is absorbed by the nucleus but in most cases the propulsion of the free neutron is powerful enough to smash the nucleus apart which produces the same outcome.

Why use Uranium for Nuclear Fission?
Uranium makes the best fissile material as it is the largest naturally occurring nucleus on earth (that hasn’t spontaneously decayed). As such it is the obvious candidate if you are trying to split minute body you would attempt with the largest minute body possible. Additionally very large nuclei require more neutrons than protons to make them stable (due to the balance between the strong nuclear force and the electromagnetic force). This is where the additional free neutrons appear from to continue the reaction and the fission of a Uranium nucleus produces an average of 2.5 free neutrons.
Difficulties with nuclear fission as an energy source
The most common isotope of Uranium is uranium-238 which makes up 99.3% of all natural Uranium but this is not a very fissile material. What is more Uranium 238 can capture the valuable neutrons becoming Uranium 239. The isotope Uranium-235 is much more fissile but makes up only 0.7% of natural Uranium. Therefore Natural Uranium must be refined to increase the percent Uranium-235 which is an expensive and tedious process.
Nuclear Fission and the environment
Energy from nuclear fission reactors is considered clean as no waste gases are produced that harm the environment. However there are concerns about the radioactive material produced, some of which have a very long half-life. When uranium-238 absorbs a neutron, for example, it produces uranium-239 which decays into plutonium-239 and has a half-life of 24,000 years.
Plutonium Fast Breeder Reactors
Plutonium -239 can itself be used to produce nuclear fission in a specially designed reactor. The plutonium is bombarded with fast moving neutrons which split the plutonium neutron. The fission of which produces on average 2.9 neutrons and again this is controlled to only allowing 1 free neutron to continue the chain reaction. This time the additional free neutrons are absorbed by uranium-239 which is used to line the reactor wall and this creates or breeds more plutonium-239 which can be used in the core of the reactor later. This type of reactor is known as a fast breeder reactor and sounds like a good option for the environment. However there are many more technical difficulties with this type of reactor and plutonium-239 from these reactors can be used, without refinement, in nuclear weapons which creates new ethical problems.