Ozone Layer
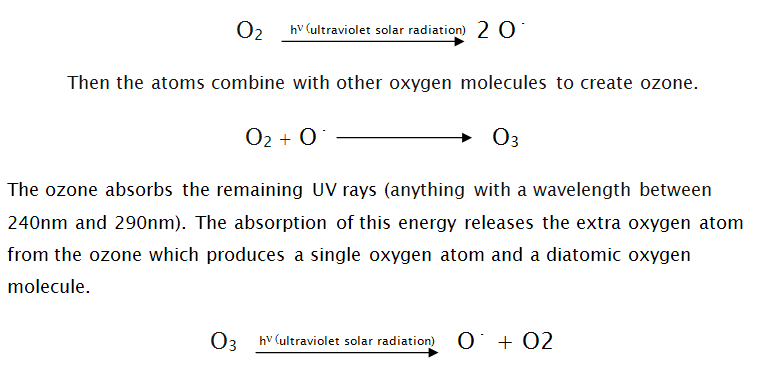
In the Stratosphere (between 10 and 50 kilometers high) is the layer containing most of the atmospheric ozone and this is mainly concentrated at about 25 kilometers up. Ozone is produced in the ozone layer mainly through ultraviolet light from solar radiation (hn) colliding with diatomic oxygen (oxygen made of two atoms). Diatomic oxygen in the stratosphere absorbs the majority of the harmful ultraviolet radiation (anything with a wavelength smaller than 240 nm) and in doing this the absorbed energy splits the molecule leaving two individual oxygen atoms.
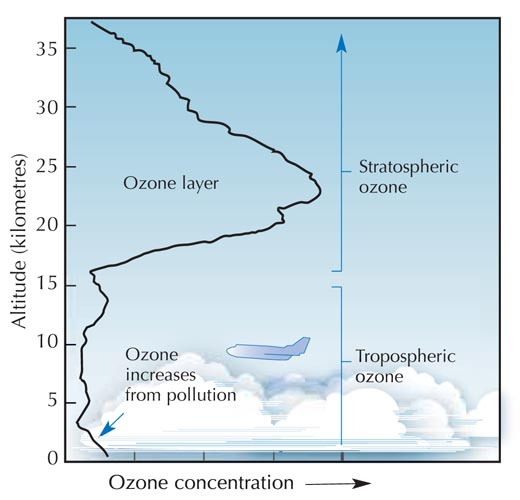
In the Stratosphere (between 10 and 50 kilometers high) is the layer containing most of the atmospheric ozone and this is mainly concentrated at about 25 kilometers up. Ozone is produced in the ozone layer mainly through ultraviolet light from solar radiation (hn) colliding with diatomic oxygen (oxygen made of two atoms). Diatomic oxygen in the stratosphere absorbs the majority of the harmful ultraviolet radiation (anything with a wavelength smaller than 240 nm) and in doing this the absorbed energy splits the molecule leaving two individual oxygen atoms.