Explore the World Through Geography, Natural Resources & Daily History
Clear, reliable and engaging guides that help you understand our planet — from UK geography education to global natural resources and On This Day history events.
Explore, discover, and learn about the wonders of our world! At Earth Site, we’re passionate about bringing geography, history, and science to life for curious minds of all ages. Whether you’re delving into historical events, uncovering the mysteries of the natural world, or seeking interactive resources, you’re in the right place.
Here, you can uncover the stories behind historical events, explore the natural wonders of our planet, and gain valuable insights into how the Earth’s systems shape our daily lives. From the towering peaks of mountain ranges to the far-reaching impacts of human innovation, we aim to make every topic both engaging and informative.
Start your journey of discovery with us today, and let’s make learning an adventure!
What We Cover
Earth Site brings together engaging and accessible educational content designed to help you understand the world, its history, and its natural systems.
🌍 Geography Education (UK & Worldwide)
We publish clear, easy-to-understand geography resources for students, teachers and curious learners. Our guides support geography education in the UK and cover physical geography, climate, ecosystems, population, and global development.
⛏️ Natural Resources & Environmental Geography
Explore detailed country profiles covering natural resources, mining, energy, geology and global environmental challenges. We show how nations manage minerals, water, land and ecosystems, and why these resources matter.
📅 On This Day in History
Every day has a story. Our On This Day history series features major events, anniversaries, traditions, and cultural milestones from around the world — with timelines, context, and fun facts.
TIMELINE
Carbon
Basic Information Discovery of Carbon Sources of Carbon Uses of Carbon Carbon Dating The Carbon Cycle Carbon Fibres Diamonds Carbon’s Cell Structure Absorption Lines of Carbon Emission Lines of Carbon Carbon (from the latin word ‘carbo’ meaning coal) Classification: Non-metal (solid at 298 K) Atomic Mass: 12.0107 g/mol Density (graphite): 2.267g/cm3 Density (Diamond): 3.513g/cm3 Colour: Diamond has no colour / graphite is black Boiling Point: 4300K (4027°C) Melting Point: 3800K (3527°C) Discovery The production of carbon in the form of charcoal has been known since records began. Sources Charcoal was produced by burning organic material in an atmosphere with reduced oxygen. Although it is possible to make superb diamonds artificially the most precious kind are produced naturally under extreme pressure over a very long period of time. Uses The ratio of carbon on earth is thought to be 99% carbon-12, just under 1% Carbon-13 and 0.0000000001% of carbon-14 with the same ratios found in all living things. Carbon-14 is an unstable isotope of carbon which can be used in the process called carbon dating. As all living things intake carbon in the above ratios, when an organism dies the Carbon 14 begins to decay. This means that over time the carbon 14 ratio decreases over time and this decrease can be measured. As scientists know the decay rate of Carbon 14 they can measure the approximate time since the organism died. Carbon is one of the fundamental building blocks of life and most carbon would have been through a process we call the Carbon Cycle. This...
Carbon Fibres
Who Created Carbon Fibres? Development of Carbon Fibres Difference between Rayon, Pitch and PAN Carbon Fibres Manufacture of Carbon Fibres PAN Carbon Fibre Production through Pyrolysis High-Strength Fibres High-Modulus Fibres and Graphitisation Creation of Carbon Fibres? The first known use of carbon fibres dates back to 1860 when an English physicist by the name of Sir Joseph Swan created the world’s first incandescent light bulb. He initially created it with a carbonized paper filament in a vacuum tube but later improved on his design in 1878 by using a carbon fibre filament derived from cotton. When Thomas Edison took out a patent for the manufacture of his light bulb in 1879 of the same design he proposed using carbonised bamboo as his filament. Edison took bamboo and heated it in a controlled atmosphere (a method known as ‘pyrolysis’ which is used in the manufacture of carbon fibres today) which produced fire-resistant carbon fibres. The fibres produced by Swan and Edison had no tensile strength but could withstand high temperatures making them perfect for use in his bulbs. In 1883 Swan produced the world’s first synthetic fibre when he squeezed nitrocellulose – obtained from tree bark- through holes to produce fibres which he could use as a new filament. This revolutionised the textile industry and led to his knighthood in 1904. All the filaments produced proved inefficient, requiring high currents to produce light and soon they were replaced with tungsten. Development of Carbon Fibres It would be around 50 years before experiments with carbon fibres would take place. In...
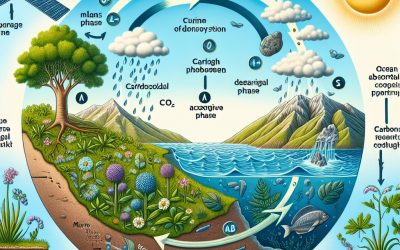
The Carbon Cycle
Plants to animals Fossil Fuels Calcium Carbonate Our planet has got many systems or cycles in place that allow for the re-use of materials which are vital for life. These include nitrogen, water and carbon. The recycling of these vital materials helps support the continued existence of life that would otherwise end. We, as all living things, do not simply use this cycle but are a part of it. Plants to animals...
Boron – Metalloid
Boron is a relatively rare metalloid both on earth and in the rest Universe. It is produced from cosmic rays and extracted from Borate minerals. Basic Information Discovery of Boron Sources of Boron Uses of Boron Boron In The Diet Use in the Nuclear Industry Use In Glass Boron’s Cell Structure Absorption Lines Emission Lines Classification: Metalloid Atomic Mass: 10.811 (7) g/mol Density: 2.34g/cm3 Colour: black Boiling Point: 4200K (3927°C) Melting Point: 2349K (2076°C) Discovery of Boron Boron (5B) compounds were used by ancient civilizations for thousands of years but the element itself wasn’t isolated until 1828. English Chemist Sir Humphrey Davy (whose assistant was Michael Faraday) originally used electrolysis to create 5B from a borates solution and named the substance boracium. Later he used boric acid combined with potassium to produce a purer form of 5B. This method was used by French chemists Joseph-Louis Gay-Lussac and Louis-Jaques Thénard Sources Commercially 5B is obtained by heating borax (Na2B4O7 10H2O) with carbon. Uses Boron can be taken as a dietary supplement and found naturally in various foods. Some evidence has shown that boron in your diet can help reduce bone loss, reduce the symptoms of osteoarthritis, improve cognative ability and cordination in older people and increase testosterone levels. There is not yet enough evidence to prove conclusively that this is the case. 5B is found naturally in fruits (such as raisins, avacado, prunes, apricots) beans and nuts (particularly brazil, hazil and cashew) or taken in supplements though anymore than 20mg a day is considered...
Beryllium: Periodic Table, Uses, Occupational Safety and Health
Beryllium: Periodic Table, Uses, Occupational Safety and Health Basic Information Discovery Sources Uses Use in Telescopes Use in Satellites Cell Structure Absorption Lines Emission Lines Beryllium (named after the mineral, beryl, that it was originally isolated from) Classification: Alkali earth metal Atomic Mass: 9.012182 (3) g/mol Density: 1.85g/cm3 Colour: grey Boiling Point: 2742K (2469°C) Melting Point: 1560K (1287°C) Beryllium: The Lightweight Power Metal Shaping Modern Industry and Safety Standards What makes beryllium so important in science, industry, and worker safety? Beryllium might not be a household name, but this light metal with atomic number 4 holds a significant place on the periodic table and in high-tech industries. Whether you’re flying on an aeroplane, undergoing an X-ray, or benefiting from advanced communication technologies, beryllium plays a hidden yet vital role. This article explores the fascinating properties of beryllium, its various applications—from alloys to nuclear reactors—and the critical occupational safety and health concerns related to its use. If you’re curious about how this relatively rare element shapes modern life, this post is a must-read. Article Outline 1. What Is Beryllium and Why Does It Matter? 2. Where Is Beryllium Found and How Is It Extracted? 3. What Are the Unique Chemical Properties of Beryllium? 4. How Is Beryllium Used in Alloys and Why? 5. How Does Beryllium Help in X-ray Technology? 6. What Role Does Beryllium Play in Nuclear Applications? 7. What Are the Health Risks of Exposure to Beryllium? 8. How Does OSHA Regulate Beryllium Exposure? 9. Why Is Beryllium So Valuable in Aerospace and Defence? 10. What Should You Know About the Future of Beryllium Use?...
Atoms
It was believed for a thousand years that atoms were the smallest component of matter, but in the late 19th century, the first evidence that atoms were made up of smaller sub-atomic particles came to light. About Atoms Diagram of an atom Electrons Discovery of Electrons Properties of Electrons Protons Discovery of Protons Properties of Protons Neutrons Discovery of Neutrons Properties of Protons About Atoms It has been known for a thousand years that all matter is made up of small particles known as atoms, but for much of this time, it was believed that atoms were the smallest component of matter. It was not until the late 19th and early 20th century that the first evidence that atoms were made up of smaller sub-atomic particles came to light. A = The Nucleus of the atom contains the protons and neutrons. Despite accounting for the majority of an atom’s mass, the nucleus occupies a minute proportion of the total space. The Diameter of the nucleus is approximately 4 femtometres or 4 x 10-15 meters. It is very difficult to measure the diameter of the nucleus because, like our atmosphere, it has no defined edge but gradually fades away. B = This is mainly just empty space where the electrons orbit the nucleus. Its diameter (and that of the whole atom) is 0.1 nanometres or 0.1 x 10-9 meters. Here is the chemical symbol for helium as you might see it on a periodic table. The number at the top is known as...
Directory
Error: Embedded data could not be displayed....